Matter exists either as in form of an element, compound or a mixture. An element is the simplest form of a substance because is made up of one type of atoms. Elements can further be classified into metals and non-metals. Compounds are formed between two or more elements that have chemically combined. Many different substances out there do exist as compounds.
A mixture is a combination of two or more substances that
have not chemically combined. This implies that they can easily be separated
using physical means. Some common methods of separating mixtures are
distillation, evaporation, filtration, chromatography, centrifugation,
magnetization, crystallisation and decanting.
(120).jpg) |
| Elements, Compounds and Mixtures |
ELEMENTS
The simplest form of a substance is called an element. It cannot be split into other simpler substances by any chemical process. An element is made up of only one kind of atom. There are 115 elements and 24 of them do not occur in nature. They form what we have as the periodic table. The elements are classified into metallic and non-metallic categories.
Metals often have high
density, high melting points, high boiling points, malleable, ductile, shiny
(lustrous), high thermal and electrical conductivity. Non-metallic elements
have low values for the stated characteristics. Each element is given a special
chemical symbol to characterize it, e.g. Carbon (C), Sulphur (S), Sodium (Na),
Potassium (K), Chlorine (CL) and Lead (Pb).
Symbols consists of one or two letters of the alphabet, the first being
capital letter- A, B, C. Several elements with the same initial letter, a
second letter or subsequent letter is added e.g. C for carbon, calcium (Ca),
Chlorine (Cl). Other use Latin names e.g. sodium (Na) from natrium, lead (Pb) from
plumbum etc
Isotopes
The number of electrons in an atom is equal to the number of protons.
The number of protons in an atom gives the atomic (proton) number for that
atom. The number of neutrons for the same element is often equal. When atoms
for an element have the same number of protons (same number of electrons), with
the same chemical properties but the number of neutrons vary, one atom type is
stable while the other atom is called an isotope for the stable atom. They
however show different physical properties.
Some examples of elements with isotopes are Cl (35, 37), H (1, 2, 3), C
(12, 13, 14), O (16, 17)
Allotropes
Some substances can crystallize in two or more forms. Some of these
substances are unstable in one form and therefore change from one form to
another under some circumstances. In situations where they change form due to
temperature, the specific temperature under which they change is called the
transition temperature. Some allotropes include
·
Calcium Carbonate (Calcite and aragonite)
·
Iodine mono-chloride (red, brown)
·
Sulphur (rhombic, monoclinic)
·
Phosphorous (red, white, black).
·
Carbon (amorphous, graphite, diamond).
·
Oxygen (02, Ozone)
·
"fin (grey, white)
Allotropes always exhibit different physical properties and at times
they may have even different chemical properties. These properties make the
allotropes be used for different purposes.
COMPOUNDS
Compounds are pure substances which are formed due to a chemical reaction between l wo or more elements. When a new substance is formed during the chemical reaction, chemical change takes place. This combining of elements can be represented by word equation: hydrogen + oxygen → water. Water molecules contain two atoms of hydrogen and one atom of oxygen and hence the chemical formula H2O.
The formula of a compound is made up from the symbols of the elements present and the ratio in which the different atoms are present. Carbon dioxide has the formula CO2. This shows that it contains one carbon atom for every two oxygen atoms. The 2 shows that there are 2 oxygen atoms present in each molecule of carbon dioxide.
Compounds are made up of fixed proportion of elements: They have a fixed proportion. Chemists call this the “Law of constant composition”. The forces that hold atoms together in compounds are called “Chemical bonds”. Reaction between elements and oxygen is called an oxidation reaction
as the substance gains oxygen. Some examples
2Mg (S) + O2(g) → 2MgO(S\
C(s) + O2(g)→ CO2(g)
When substances react such that there is a loss of oxygen, the process
is called reduction. Some examples:
CuO (S) + H2(g) →Cu(S)
+ H20(g).
Fe2O3(S) + 3CO(g) → 2Fe(S)
+ 3CO2(g)
Look at these REDOX reactions
1. This is an
example of oxidation reaction
Carbon + Oxygen → Carbon dioxide
C(s) + O2(g) → CO2(g)
Oxidation reaction is gain of oxygen, loss of hydrogen or electrons.
2. This is an
example of a reduction reaction
Copper Oxide + Hydrogen → Copper + Water
CuG(s) + H2(g)
→ Cu (S) + H2O(g)
deduction reaction is loss of oxygen, gain of hydrogen or electron.
MOLECULES
The atoms are joined together
in small groups. These small groups are called “molecules”. For example
the atoms of Hydrogen, Iodine, Chlorine, Nitrogen, Oxygen, Fluorine and Bromine
are each joined in pairs and are known as “diatomic”
molecules, Phosphorus atoms are joined in four and sulphur in eight. In
chemical shorthand the molecule of Chlorine is written s Cl2.
The gaseous element helium,
neon, argon, krypton, Xenon and Radon are composed of separate and individual
atoms. They are “monatomic”; they
are shorthanded Ne, Ar, Kr, Xe and Rn respectively.
Molecules are not always
formed by atoms of the same type joining together. For example, water exists as
molecule containing Oxygen and hydrogen atoms.
MIXTURES
Mixtures contain more than one substance (element or compound). The substances of the mixture have their specific physical properties. Our world is very complex, owing to the vast range of pure substances available, and to the variety of ways in which these pure substances can mix with each other. In everyday life we do not ‘handle’ pure substances very often.
The air we breathe is not a single, pure substance - and we could not live in it if it were! Water would be rather tasteless if we drank it distilled - indeed, some companies do market distilled water that has had salts added back to it.
It is therefore possible to separate these substances provided there are suitable differences in their physical properties. If the mixture is made to undergo a chemical reaction, then separation thereafter becomes more complicated.
Types of mixtures
Mixtures may be Homogeneous or Heterogeneous
• A homogeneous mixture has the same composition of elements/or compounds throughout - evenly distributed. It is also called solution. An example of such a mixture is copper II sulphate solution.
A heterogeneous has different regions or areas with different compositions and properties - not evenly distributed. An example of such a mixture is concrete.
Each mixture must be made from at least two parts, or phases, which may be solid, liquid or gas.
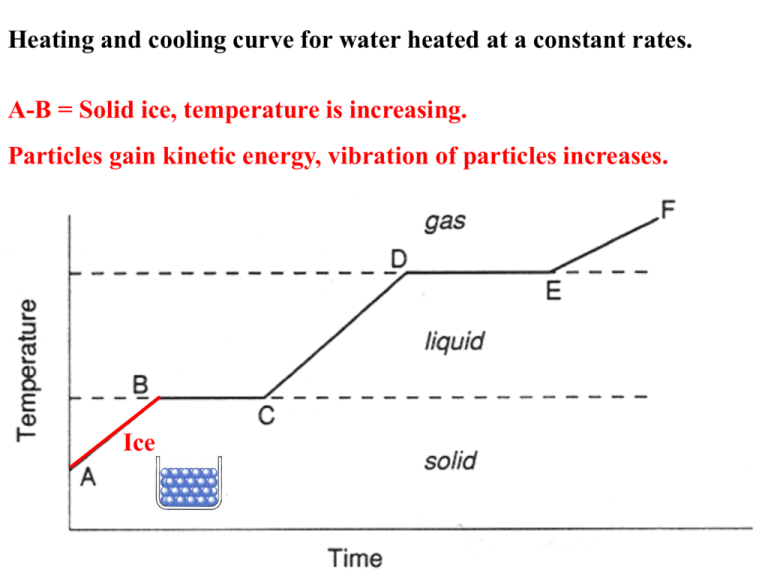 |
| Element, Compound and Mixtures - Cooling curve |
The temperature stays constant while the gas condenses, and while the liquid freezes. A cooling mixture of ice and salt could be used to lower the temperature below 0 °C.T
There are a number of
different ways in which the three states can be combined. In some, the states
are completely mixed to become one single state or phase - ‘you cannot see the
join’. This is a homogeneous mixture. Technically, the term ‘solution’ is used
for this type of mixture. Solid salt dissolves in liquid water to produce a
liquid mixture - a salt solution. In other types of mixture, the
states remain separate (a heterogeneous mixture). One phase is
broken up into small particles, droplets, or bubbles, within the main phase.
Solutions and homogeneous mixtures
Table 2.3 shows us some of the
ways in which substances in different states can combine. Perhaps the most
important idea here is that of one substance dissolving in another - the idea
of a solution.
We most often think of a solution as being made of a solid dissolved in a
liquid. Two- thirds of the Earth’s surface is covered by a solution of various
salts in water. The salts are totally dispersed into the water and cannot be
seen. However, other substances that are not normally solid are dissolved in
sea water. For example, the dissolved gases, oxygen and carbon dioxide, are
important for life to continue in the oceans.
Less obvious perhaps, but
quite common, are solutions of one liquid in another. Alcohol mixes (dissolves) completely with water. Beer,
wine and whisky do not separate out into layers of alcohol and water (even when
the alcohol content is quite high). Alcohol and water are completely miscible:
they make a solution.
Table 2.3 Important examples
of different types of mixture
|
Type of mixture |
Mixture |
Description |
Examples |
|
Homogeneous
mixtures (solutions) |
Solution
* of solid in liquid |
transparent
solution of solid dissolved in liquid |
sea
water, sugar in water, salt solutions |
|
Solution of two miscible liquids |
single
layer of transparent liquid |
vodka
(alcohol and water), two- stroke motor oil (petrol and oil) |
|
|
Solution of gas in liquid |
transparent
solution of gas dissolved in a liquid |
mineral
and soda waters, champagne, ‘fizzy’ soft drinks |
|
|
|
Mixture
of gas in gas |
transparent
mixture of two or more gases |
air |
|
Alloy of
two solid metals |
solid,
evenly spread mixture of two metals |
brass,
bronze |
|
|
Heterogeneous
mixtures |
Suspension
of solid in liquid |
cloudy
mixture of solid particles suspended in a liquid |
river
water carrying mud and silt, flour in water, kaolin indigestion medicine |
|
Gel |
jelly-like
mixture of solid and liquid, liquid trapped in the solid |
fruit jelly,
agar gel |
|
|
Emulsion
of two immiscible liquids |
cloudy
mixture of tiny drops of one liquid suspended in another liquid |
skin
cream, milk, salad dressing, mayonnaise |
|
|
Aerosol
of either a liquid or solid in a gas |
small
droplets of liquid, or particles of solid, dispersed in a gas |
liquid-in-gas:
mist, clouds, paint spray solid-in-gas:
smoke, dusty air |
|
|
Foam of
gas in liquid |
many
small bubbles of gas trapped in liquid |
washing
lather, froth or ‘head’ on beer, shaving foam |
|
|
Solid
foam of gas in solid |
many
small bubbles of gas trapped in a solid |
polystyrene
foam, foam rubber, bread, sponge cake |
These solutions are particularly important in chemistry.
Technically the air itself
could be described as a solution of several gases in nitrogen, though this would
be an unusual everyday use of the term. However, it is interesting to note that
different gases always mix completely with each other. Likewise, alloys are
homogeneous mixtures of metals, though we do not usually call them solutions.
They are made by mixing the liquid metals together (dissolving one metal in the
other) before solidifying the alloy.
Suspensions and heterogeneous mixtures
There are a great number of
combinations of substances that do not mix: solids that do not dissolve in
water; liquids that do not mix with each other (they are immiscible). Flour does
not dissolve in water but forms a slurry or suspension. The particles
of solid are simply dispersed (spread) throughout the water and will eventually
settle out if left to stand. Sand does not dissolve in water either. The ocean
tides shift and deposit it on beaches and sandbars throughout the world. The
great estuaries such as the Nile or Mississippi deltas are silted up by
suspended soil deposited from the river.
Oil and water do not mix. Salad dressing made from olive oil and vinegar (a mixture of ethanoic acid and water) will settle out into two layers. When shaken, it forms an emulsion with droplets of oil suspended throughout the vinegar. Unlike pure liquids, emulsions are cloudy (opaque) so you cannot see through them. To stop an emulsion, such as mayonnaise, separating out into layers, an emulsifier is added.
This prevents
the small droplets of oil coming together to form larger droplets and then a
separate layer. In traditional mayonnaise the emulsifier is egg yolk, which
contains lecithin. Other food products contain other emulsifiers. In the
European system for labelling food additives, emulsifiers are given E numbers
from E322 to E494. E322 is lecithin.
The world of cosmetics is full
of examples of emulsions. Skin moisturising creams are emulsions of oils in
water; the oils prevent the skin from drying out. It is possible to switch the
balance of the two phases in a cosmetic preparation. Cold cream and cleansing
cream are emulsions of water in oils. They have an oilier ‘feel’ or texture than
hand or face creams.
There are
two ways in which mixtures can be formed between different substances:
• homogeneous mixtures, where the
substances are totally mixed together and are indistinguishable - examples
include solutions of salts and sugars in water.
• heterogeneous mixtures, where the
substances remain separate and one substance is spread throughout the other as
small particles, droplets, or bubbles - examples include suspensions of
insoluble solids in water.
Differences between Mixtures and Compounds
Mixture | Compound |
The constituent substances can be separated from one another by physical methods | The constituent elements cannot be separated by physical method, chemical reaction is necessary. |
Mixture may vary widely in composition | Compounds are fixed in their compositions by mass of the elements present |
Mixing is not usually accompanied by external effects such as explosion, evolution of heat or volume change for gases | Chemical change is usually accompanied by one or more of these effects |
The properties of the mixture are the sum of the properties of the constituents of the mixture. | The properties of a compound are peculiar to it and are usually very different from the properties of the constituent elements. |
Colloids and suspensions
The heterogeneous mixtures
listed in table 2.3 can be divided into two types - suspensions and colloids.
The difference depends on the size of the particles suspended, or dispersed, in
the surrounding medium.
• Suspensions contain relatively large particles (over l000nm, where
1 nm = 10-9m) of an insoluble solid, or droplets of an insoluble
liquid. In time, the particles or droplets settle out.
• Colloids contain smaller particles (1-1000nm) and take various
forms. Emulsions, sols, gels, aerosols and foams are all examples of colloids.
The particles of a colloid are too small to be seen by eye, but they do scatter
light.
Colloids fill an important place in our lives. Our blood contains proteins spread throughout a watery medium; milk is a complex colloid containing fat droplets and proteins dispersed in water. We also manufacture a large number of colloids to serve a wide range of purposes. ‘Emulsion’ paint is an example of a colloidal system.
In white ‘emulsion’ paint, the dispersed particles are solid (for example
titanium(iv) oxide particles), mixed up in water but not dissolved in it. A
true emulsion is made from one liquid dispersed in another. ‘Emulsion’ paint is
not in fact a true emulsion. Nor is the photographic ‘emulsion’ coated on
films. The light-sensitive layer of a film consists of fine crystals of silver
bromide trapped in a gelatine gel.
Every
colloid has at least two parts :
•
the dispersed
phase is split into very small particles - for example the fat in milk or
the water droplets in mist.
•
the continuous
phase contains the dispersed phase spread throughout it - for example the
water in milk or the air in mist.
Table 2.4 The dispersed and
continuous phases of different types of colloid
|
Dispersed
phase |
Continuous
phase |
Type of
colloid |
Example |
|
liquid |
gas |
aerosol |
mist |
|
solid |
gas |
aerosol |
smoke |
|
gas |
liquid |
foam |
Shaving
cream |
|
liquid |
liquid |
emulsion |
face
cream |
|
solid |
liquid |
sol |
paint |
|
gas |
solid |
foam |
sponge |
|
liquid |
solid |
gel |
butter |











0 Comments